Published On January 23, 2017
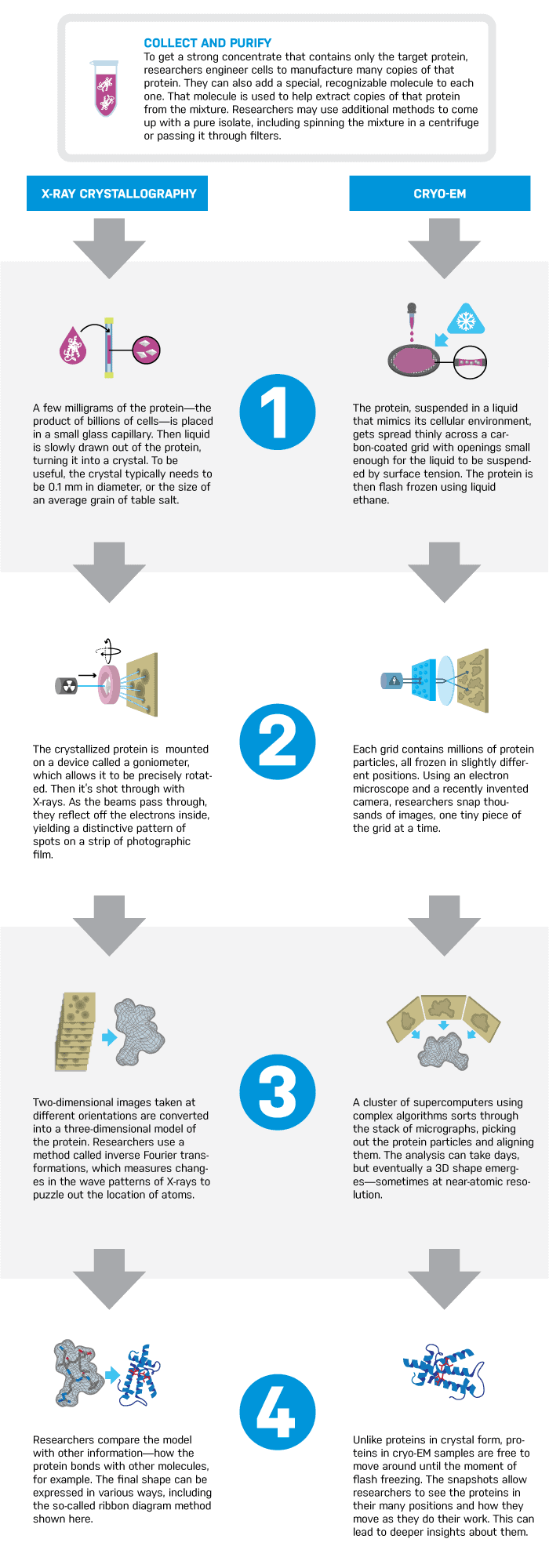
Proteins are the workhorses of the body. But they’re so small—just a few nanometers in length—that it’s immensely difficult to see them. That’s a problem, because knowing the structure of a protein can be crucial in determining how to lock on to its shape and influence its behavior. Using the shapes of about 100,000 proteins that have currently been mapped, researchers have already developed hundreds of drugs.
About 90% of those proteins have been mapped through a process called X-ray crystallography. But not all proteins are good candidates, and holdouts include critical membrane proteins, which reside in a cell’s outer layer, and which are the targets of a lion’s share of drugs. Only 500 such protein maps exist.
Refinements in a method called cryo-EM (cryo-electron microscopy), however, offer a promising new window into the shape of proteins. Cryo-EM images are gaining in resolution, and can also capture proteins in a range of positions that crystallography may miss.
Stay on the frontiers of medicine
Related Stories
- Crystal in the Sky
The best place to observe the building blocks of the human body might be in outer space.
- Origami Medicine
When proteins misfold, molecular chaos ensues, leading to cystic fibrosis and other ills. New research aims to unwind the mistakes.
- Tangled Up in Tau
Trouble with the protein may underlie most kinds of dementia, potentially including Alzheimer’s. New drugs could help.